
From Central and Eastern Europe to Switzerland, we are building regulatory-compliant, sustainable and long-term business partnerships.
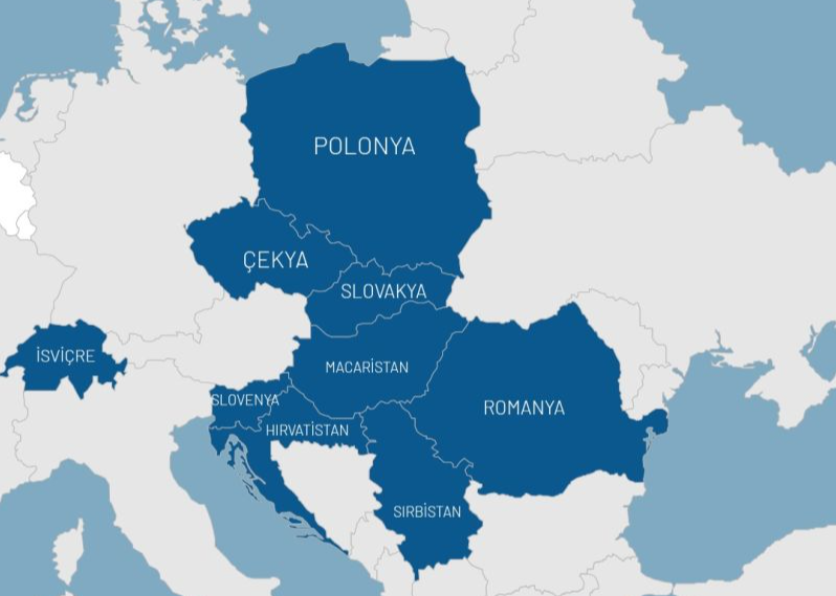
With strong regional partnerships and flexible distribution models, Mealis operates in European markets with different regulatory structures.
Our medical device, pharmaceutical and nutraceutical product portfolio is positioned in line with the requirements of local authorities, patient needs and clinical practice.
Strong regulatory compliance
Full compliance with EMA, local agencies and national legislation.
Local business partnerships
Strong, experienced distributor networks in every country.
We are present in the following countries through distributorships, license agreements or strategic partnerships.
Central Europe
Strong access through regional hubs and an extensive pharmacy network.
Central Europe
Integrated marketing and sales structures thanks to the twin market structure.
Central Europe
Sustainable growth through long-term distributorship agreements.
Eastern Europe
Access to public and private hospitals across a wide geography.
Adriatic Region
High value-added positioning with a niche product portfolio.
Balkans & Western Europe
Flexible distribution models compatible with different regulatory regimes.
You can contact us for detailed information about cooperation or distributorship opportunities in international markets.
Contact Us for Collaboration